Research Interest
Our lab is interested in building technologies that solve critical research bottlenecks and answer fundamental biological questions. Below you'll see a few of our ongoing projects and their applications.
Combining high-throughput protein characterization with protein language models.
While comprehensive mutagenesis studies have provided unprecedented insight to how proteins work, they are highly labor intensive, which means only a small number of proteins have been subject to this comprehensive analysis. To overcome this experimental limitation, our team is pioneering the use of protein language models coupled with high-throughput functional assays to rapidly decipher protein fitness landscapes. We are interested in applying this approach to guide the development of designer therapeutic proteins and predict the effects of clinical variants on protein function.
Combinatorial protein engineering
Our lab previously developed a series of potent second generation Cas9-based transcriptional modulators. To date, these reagents have been requested by over 1,000 labs across the globe. In ongoing work, we have developed a novel method of screening combinatorial libraries of thousands of Cas9 variants to identify a third generation of Cas9 activators and repressors. Newly established effectors will be used to perform CRISPR screens to identify regulators of neuroinflammation and tumor cell growth. We are also adapting our regulators for in vivo applications within animal models of disease.
In vivo CRISPR screens
While CRISPR has revolutionized how we interrogate biological systems, to date much of the work comes from studying cells in isolation, outside of their physiologic environment. We have assembled an interdisciplinary team of scientist specializing in technology development, high-throughput screening, and neurobiology to perform in vivo CRISPR screens within animal models of neurodegeneration. By identifying genes which influence neuronal survival within neurodegenerative disease states, we aim to better understand the mechanisms underlying the profound neuronal loss seen within patients, along with identifying novel points for therapeutic intervention.
Guide+Donor libraries for high-throughput functional genomics
Our team has developed a strategy to generate thousands of yeast mutants, each with a defined genetic alteration (point mutation, deletion, or insertion) within a single reaction. We have also created a pipeline for simultaneously tracking the fitness of all of the resulting mutants en masse. We are applying our technology to gain insights into the coding, and non-coding portions of the genome, optimize metabolic pathways and better understand how point mutations influence protein structure and function.
Building technologies to speed the rate of antiviral drug discovery
Expanding upon our lab’s expertise in assay development, we have created a series of technologies to help increase the pace of antiviral drug discovery. These innovations have ranged from devising a method to screen for viral protease inhibitors in mammalian cells without requiring the use of live virus, to generating a novel multiplex drug screening approach that lets us search for inhibitors against dozen of viral proteins at once. Our long-term goal is to leverage these technologies to not only address the ongoing COVID-19 pandemic but also to help fill the numerous gaps within our existing antiviral armament.
DNA barcoded libraries for understanding regulators of neurodegeneration
By employing DNA barcodes, we are able to track the growth of dozens of cellular models at a time. We use these pools of barcoded cells to perform multiplexed screens aimed at understanding the genes involved in tolerance to misfolded proteins and investigating drugs that rescue various cell-based models of neurodegenerative disease. By using DNA-barcoding we are able to improve screening throughput by over an order of magnitude compared to conventional approaches, enabling us to obtain much richer datasets.
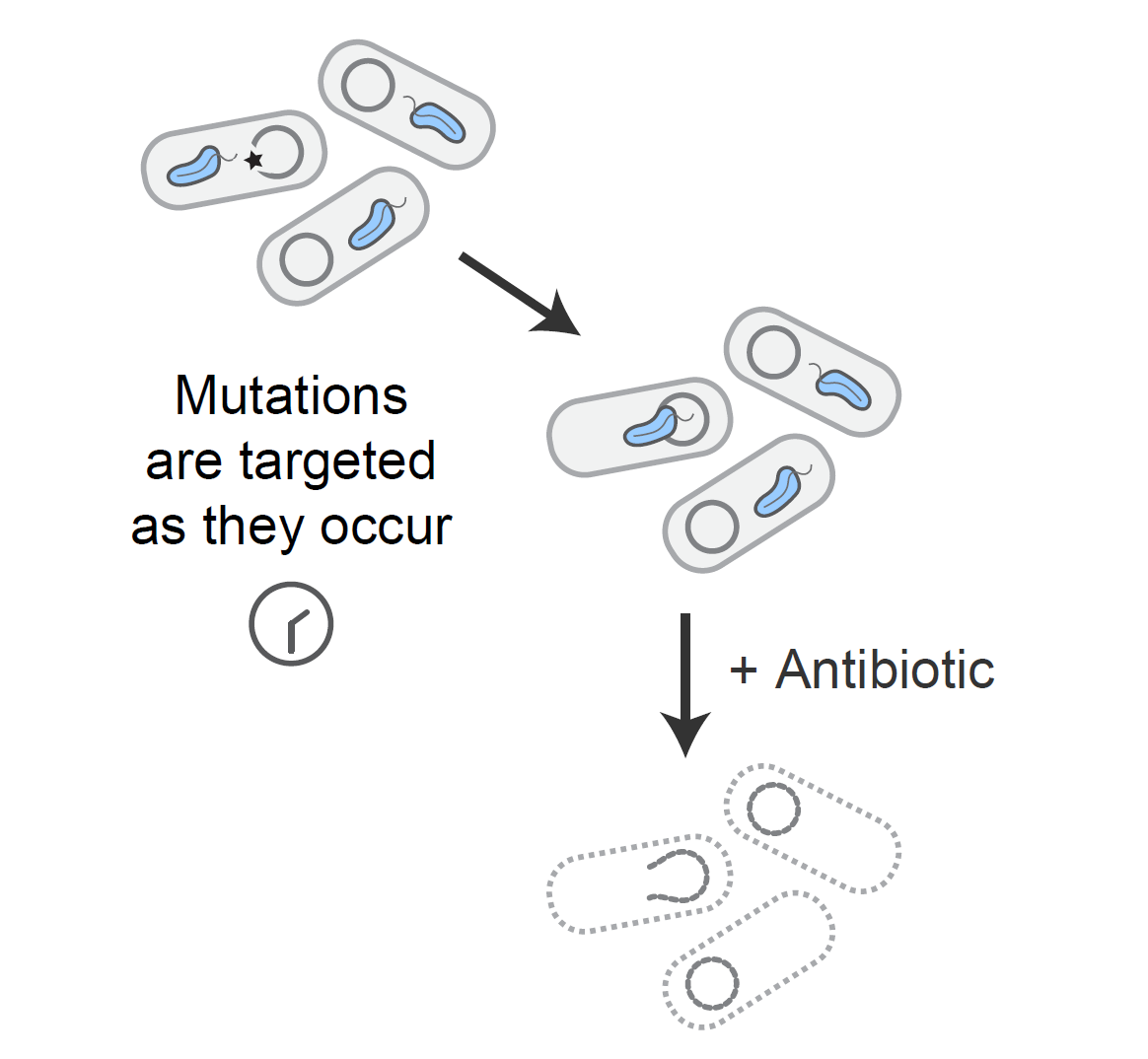
Tuned guide RNAs to control evolution
We developed a system to endow Cas9 with single nucleotide specificity through the development of tuned guide RNAs. With this method in hand, we developed a "genomic surveillance system" which prevents cells from gaining undesired mutations. We are exploring uses of this technology to control evolutionary trajectories to enrich for unconventional solutions to a given selection pressure.
Synthetic gene drives to bias inheritance and aid in the dissection of fundamental biological processing
Gene drives are selfish genetic elements that "cheat" Mendelian inheritance. We were among the first to generate synthetic gene drives using Cas9. Our team is using this tool to perform rapid combinatorial knockouts within the clinically relevant fungal pathogen Candida albicans to understand the cellular mechanisms regulating pathogenicity.